 
Both enthalpy and entropy conspire to reduce the acidity of H-F. 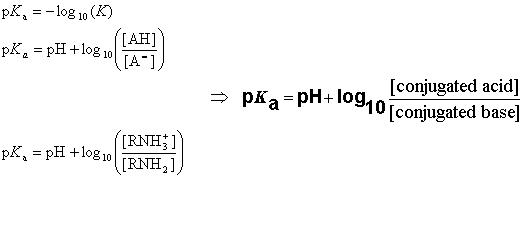
In liquid anhydrous HF, self-ionization occurs: 3 HF H 2 F + + HF 2 Even though it is highly corrosive, hydrofluoric acid is not considered to be a strong acid because it does not completely dissociate in water. In cell biology, protein kinase A (PKA) is a family of enzymes whose activity is dependent on cellular levels of cyclic AMP (cAMP). This generally means that in aqueous solution at standard temperature and pressure, the concentration of hydronium ions is equal to the concentration of strong acid introduced to the solution. It is used to determine how much an acid dissociates in solution. The lesser the pKa is, the molecule would hold proton less tightly hence the more potent the acid will be. 6.3 An example is hydrochloric acid (HCl), whose pKa is -6.3. What is pKa pKa is the criterion used to determine the acidity of the molecule. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
